
-
 Top Russian official in Washington for talks on improving ties
Top Russian official in Washington for talks on improving ties
-
Sinner's former physio to blame for failed dope tests, says ex-physical trainer

-
 Germany slams Trump tariffs, US tech titans in crosshairs
Germany slams Trump tariffs, US tech titans in crosshairs
-
Trump tariff blitz sparks retaliation threats, economic fears

-
 Search for Malaysia's long missing MH370 suspended
Search for Malaysia's long missing MH370 suspended
-
Hungary announces ICC withdrawal as Israel's Netanyahu visits

-
 Trump's tariffs sting Asian giants, including US allies
Trump's tariffs sting Asian giants, including US allies
-
India says 'examining the implications' of US tariffs

-
 Evenepoel set to make injury return at Tour de Romandie
Evenepoel set to make injury return at Tour de Romandie
-
USA sole bidder for 2031 Women's World Cup, UK set to host in 2035 - Infantino

-
 McLaren's Norris says it's 'our turn' for success
McLaren's Norris says it's 'our turn' for success
-
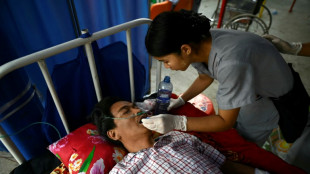
Lessons and liquids: buried alive in Myanmar's earthquake
-
 Trump tariffs spark fears for Asian jobs, exporting sectors
Trump tariffs spark fears for Asian jobs, exporting sectors
-
Stocks and dollar sink, havens rally as Trump tariffs fan trade war

-
 Runners fly to North Korea for first post-Covid Pyongyang Marathon
Runners fly to North Korea for first post-Covid Pyongyang Marathon
-
Hamilton rubbishes claims he's lost faith in Ferrari

-
 Nintendo Switch 2 sparks excitement despite high price
Nintendo Switch 2 sparks excitement despite high price
-
Sri Lanka's crackdown on dogs for India PM's visit sparks protest

-
 S Korea police raise security levels ahead of impeachment verdict
S Korea police raise security levels ahead of impeachment verdict
-
China vows 'countermeasures' to sweeping new US tariffs

-
 Trump jolts allies, foes and markets with tariff blitz
Trump jolts allies, foes and markets with tariff blitz
-
France says EU to target US online services after Trump tariffs

-
 Tsunoda vows to bring 'something different' after Red Bull promotion
Tsunoda vows to bring 'something different' after Red Bull promotion
-
Verstappen not happy with Tsunoda-Lawson Red Bull swap

-
 Experts accuse 54 top Nicaragua officials of grave abuses
Experts accuse 54 top Nicaragua officials of grave abuses
-
Remains of 30th victim of Los Angeles fires found

-
 EU to target US online services after Trump tariffs: France
EU to target US online services after Trump tariffs: France
-
How Trump's 'liberation day' tariffs will impact China

-
 Malaysia suspends search for long-missing flight MH370
Malaysia suspends search for long-missing flight MH370
-
Search for long-missing flight MH370 suspended: Malaysia minister

-
 Europe hits out at Trump tariffs, keeps door open for talks
Europe hits out at Trump tariffs, keeps door open for talks
-
Myanmar's junta chief to head to Bangkok summit as quake toll surpasses 3,000

-
 Lawson vows to prove he belongs in F1 after shock of Red Bull axing
Lawson vows to prove he belongs in F1 after shock of Red Bull axing
-
Australia sweats through hottest 12 months on record: official data

-
 Livestock theft is central to jihadist economy in west Africa
Livestock theft is central to jihadist economy in west Africa
-
South African artist champions hyenas in 'eco-queer' quest

-
 Danish PM in 'unity' Greenland visit amid US takeover threats
Danish PM in 'unity' Greenland visit amid US takeover threats
-
Taiwan says US tariffs 'highly unreasonable'

-
 Lawson says ruthless Red Bull axing was 'tough to hear'
Lawson says ruthless Red Bull axing was 'tough to hear'
-
Heat humble Celtics for sixth straight win, Thunder roll on

-
 Trump escalates trade war with sweeping global tariffs
Trump escalates trade war with sweeping global tariffs
-
Japan says US tariffs 'extremely regrettable', may break WTO rules

-
 South Koreans anxious, angry as court to rule on impeached president
South Koreans anxious, angry as court to rule on impeached president
-
Juve at in-form Roma with Champions League in the balance

-
 Injuries put undermanned Bayern's title bid to the test
Injuries put undermanned Bayern's title bid to the test
-
Ovechkin scores 892nd goal -- three away from Gretzky's NHL record

-
 Australian former rugby star Petaia signs for NFL's Chargers
Australian former rugby star Petaia signs for NFL's Chargers
-
China says opposes new US tariffs, vows 'countermeasures'

-
 Athletics world watching as 'Grand Slam Track' prepares for launch
Athletics world watching as 'Grand Slam Track' prepares for launch
-
Heat humble Celtics for sixth straight win, Cavs top Knicks


ClearPoint Neuro Announces FDA Clearance for ClearPoint Navigation Software Version 3.0
SOLANA BEACH, CALIFORNIA / ACCESS Newswire / January 27, 2025 / ClearPoint Neuro, Inc. (Nasdaq:CLPT) (the "Company"), a global device, cell, and gene therapy-enabling company offering precise navigation to the brain and spine, today announced it has received 510(k) clearance for its ClearPoint Navigation Software Version 3.0.
"As we prepare for the wave of new patients that will be treated with cell and gene therapies in the years ahead, it is crucial that ClearPoint helps healthcare providers be ready by providing simplified workflows, offering solutions to increase surgical capacity and expanding access to our hardware and software," commented Joe Burnett, President and CEO at ClearPoint Neuro. "The newest ClearPoint 3.0 platform is designed to do exactly that by offering one of the fastest and most accurate navigation systems available while enabling the flexibility to be used in both the MRI suite and the operating room. Today, more than nighty-five percent of all stereotactic procedures in the United States take place in the operating room using Computed Tomography (CT) as the imaging modality. Using CT, the ClearPoint Software now supports the entire operating room procedure, eliminating the need for additional third-party navigation systems. Hospitals can use ClearPoint in either or both settings, benefiting from a consistent workflow that streamlines procedures and builds proficiency. We believe that this single, flexible solution will benefit our 50+ global biopharma partners with the potential to standardize cell and gene therapy delivery around the world, starting here in the United States."
ClearPoint Navigation Software Version 3.0 introduces an intraoperative CT workflow that builds on over a decade of experience in enabling MRI-guided stereotactic procedures. While previous versions of ClearPoint software supported MRI-guided workflows exclusively, the latest release extends ClearPoint navigation capabilities to the operating room. With compatibility for intraoperative CT and Conebeam CT imaging, this enhancement broadens access to precision-guided neurosurgery for facilities without intraoperative MRI capabilities. The ClearPoint Navigation Software Version 3.0 when used in conjunction with the SmartFrame® XG stereotactic frame, is intended to provide precise stereotactic guidance when placing instruments or devices during neurosurgical procedures. These procedures include biopsies, catheter and electrode insertion including deep brain stimulation (asleep or awake) lead placement.
The Company plans to initiate a limited market release for ClearPoint Navigation Version 3.0 in Q1 2025, with a planned full market release in the second half of 2025.
About ClearPoint Neuro
ClearPoint Neuro is a device, cell, and gene therapy-enabling company offering precise navigation to the brain and spine. The Company uniquely provides both established clinical products as well as pre-clinical development services for controlled drug and device delivery. The Company's flagship product, the ClearPoint Neuro Navigation System, has FDA clearance and is CE-marked. ClearPoint Neuro is engaged with healthcare and research centers in North America, Europe, Asia, and South America. The Company is also partnered with the most innovative pharmaceutical/biotech companies, academic centers, and contract research organizations, providing solutions for direct CNS delivery of therapeutics in pre-clinical studies and clinical trials worldwide. To date, thousands of procedures have been performed and supported by the Company's field-based clinical specialist team, which offers support and services to our customers and partners worldwide. For more information, please visit www.clearpointneuro.com.
Forward-Looking Statements
This press release contains forward-looking statements within the context of the federal securities laws, including the Company's expectation for the future market of its products and services, and other performance and results. These forward-looking statements are based on management's current expectations and are subject to the risks inherent in the business, which may cause the Company's actual results to differ materially from those expressed in or implied by forward-looking statements. Particular uncertainties and risks include those relating to: global and political instability, supply chain disruptions, labor shortages, and macroeconomic and inflationary conditions; future revenue from sales of the Company's products and services; the Company's ability to market, commercialize and achieve broader market acceptance for new products and services offered by the Company; the ability of our biologics and drug delivery partners to achieve commercial success, including their use of the Company's products and services in their delivery of therapies; the Company's expectations, projections and estimates regarding expenses, future revenue, capital requirements, and the availability of and the need for additional financing; the Company's ability to obtain additional funding to support its research and development programs; the ability of the Company to manage the growth of its business; the Company's ability to attract and retain its key employees; and risks inherent in the research, development, and regulatory approval of new products. More detailed information on these and additional factors that could affect the Company's actual results are described in the "Risk Factors" section of the Company's Annual Report on Form 10-K for the year ended December 31, 2023, and the Company's Quarterly Report on Form 10-Q for the three months ended September 30, 2024, both of which have been filed with the Securities and Exchange Commission, and the Company's Annual Report on Form 10-K for the year ended December 31, 2024, which the Company intends to file with the Securities and Exchange Commission on or before March 31, 2025. The Company does not assume any obligation to update these forward-looking statements.
Contact Information
Danilo D'Alessandro
Chief Financial Officer
[email protected]
(888) 287-9109 ext. 3
Media Contact
[email protected]
SOURCE: ClearPoint Neuro, Inc.
View the original press release on ACCESS Newswire
Y.Kobayashi--AMWN



