-
 Academy to host first overseas ceremony to honor young filmmakers
Academy to host first overseas ceremony to honor young filmmakers
-
No doctor necessary: US okays nasal spray flu vaccine for self-use
-
 Gurbaz, birthday boy Rashid lead Afghanistan to 177-run rout of South Africa
Gurbaz, birthday boy Rashid lead Afghanistan to 177-run rout of South Africa
-
Former delivery man Baldwin leads star names at PGA Championship

-
 Trump shooting: Secret Service admits complacency
Trump shooting: Secret Service admits complacency
-
Can an ambitious Milei make Argentina an AI giant?

-
 Haiti, its suffering growing, in 'race against time': UN expert
Haiti, its suffering growing, in 'race against time': UN expert
-
Ibrahim Aqil, the Hezbollah elite unit commander wanted by the US

-
 Chinese forward Cui signs NBA contract with Brooklyn Nets
Chinese forward Cui signs NBA contract with Brooklyn Nets
-
US Fed dissenter calls for 'measured' pace of rate cuts

-
 Guardiola tells players to lead change over workload as Kompany demands cap on games
Guardiola tells players to lead change over workload as Kompany demands cap on games
-
Norway limits wild salmon fishing as stocks hit new lows

-
 Top Hezbollah commander killed in Israeli strike on Beirut
Top Hezbollah commander killed in Israeli strike on Beirut
-
Rotterdam fatal knife attacker suspected of 'terrorist motive'

-
 First early votes cast in knife-edge US presidential election
First early votes cast in knife-edge US presidential election
-
Top-ranked Swiatek out of Beijing due to 'personal matters'

-
 Hard-right Reform UK looks to the future after vote success
Hard-right Reform UK looks to the future after vote success
-
Embiid agrees to NBA contract extension with 76ers

-
 Joshua aims to complete road to redemption in Dubois bout
Joshua aims to complete road to redemption in Dubois bout
-
World champion Bagnaia sets pace with lap record at Misano

-
 Biden says 'working' to get people back to homes on Israel-Lebanon border
Biden says 'working' to get people back to homes on Israel-Lebanon border
-
Pope criticises Argentina's crackdown on protesters

-
 Court limits screenings of videos in France mass rape case
Court limits screenings of videos in France mass rape case
-
Gurbaz century takes Afghanistan to 311-4 in 2nd ODI

-
 Central banks face 'difficult balancing act': IMF chief
Central banks face 'difficult balancing act': IMF chief
-
McLaren's Norris sets Singapore pace as struggling Verstappen 15th

-
 Guardiola tells players to lead change over workload fears
Guardiola tells players to lead change over workload fears
-
Paris Olympics sports equipment moves to new homes

-
 'Happy' Kinghorn relishing life at Toulouse
'Happy' Kinghorn relishing life at Toulouse
-
Norris sets Singapore pace as Verstappen only 15th

-
 8 dead in Israeli strike, source says Hezbollah commander killed
8 dead in Israeli strike, source says Hezbollah commander killed
-
Germany to bid to host women's Euro 2029

-
 Portugal brings deadly forest fires under control
Portugal brings deadly forest fires under control
-
Postecoglou defends Solanke after slow start to Spurs career

-
 US nuclear plant Three Mile Island to reopen to power Microsoft
US nuclear plant Three Mile Island to reopen to power Microsoft
-
Arteta urges Arsenal to take next step in Man City showdown

-
 Stock markets fall after Fed-fuelled rally
Stock markets fall after Fed-fuelled rally
-
Top Hezbollah commander 'killed' in Israel strike

-
 Poland charges Russian over attack on Navalny ally: prosecutors
Poland charges Russian over attack on Navalny ally: prosecutors
-
Man City have rest 'advantage' in Arsenal showdown: Guardiola

-
 Maresca has 'no doubt' in Jackson as Chelsea's number nine
Maresca has 'no doubt' in Jackson as Chelsea's number nine
-
EU chief announces 35 bn euro loan plan for Ukraine before winter

-
 From TikTok to Hollywood, the irresistible rise of Italy's Khaby Lame
From TikTok to Hollywood, the irresistible rise of Italy's Khaby Lame
-
Verstappen punished for swearing in Singapore press conference

-
 Sri Lanka lead by 202 in first New Zealand Test
Sri Lanka lead by 202 in first New Zealand Test
-
Brook 'not too fussed' by England's batting in heavy Australia loss

-
 India's Ashwin 'happy' to embrace pressure
India's Ashwin 'happy' to embrace pressure
-
A modern 'Trojan Horse': two days of mayhem in Lebanon

-
 Third of Burundi mpox cases in children under five: UN
Third of Burundi mpox cases in children under five: UN
-
Man Utd appoint Foster + Partners to develop Old Trafford 'masterplan'


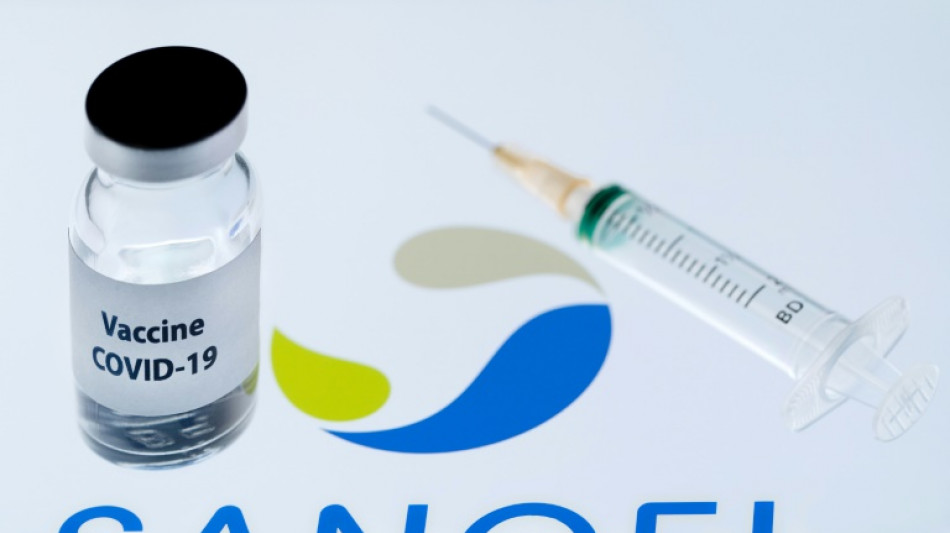
EU watchdog backs Sanofi Covid booster jab
The EU on Thursday approved a Covid booster vaccine by French drug maker Sanofi and Britain's GSK after it gave positive results against the Omicron variant in trials.
The approval of the "next generation" jab by the European Medicines Agency (EMA) is a shot in the arm for Sanofi and GSK, which have lagged behind rivals in offering a vaccine.
The VidPrevtyn Beta jab could be used as a booster in adults previously given mRNA jabs like those from Pfizer/BioNTech and Moderna, or viral vector vaccines made by AstraZeneca and Johnson & Johnson, the EMA said.
"A booster dose of VidPrevtyn Beta is expected to be at least as effective as Comirnaty (Pfizer's vaccine) at restoring protection against Covid-19," the Amsterdam-based EMA said.
A trial of 162 adults given the Sanofi-GSK booster showed that it triggers a higher production of antibodies against the Omicron BA.1 subvariant than Pfizer's jab, the regulator said.
A second study restored immunity in 627 adults who received other vaccines for their first course of jabs.
Sanofi said it was ready to start its first shipments of the booster in Britain and the EU, in line with advance contracts for 70 million doses.
"Today's approval validates our research in developing a novel solution for the Covid-19 pandemic," Thomas Triomphe, Sanofi executive vice president for vaccines, said.
The vaccine combines a Sanofi-developed antigen based on the Beta variant, which stimulates the production of germ-killing antibodies, with GSK's adjuvant technology, a substance that bolsters the immune response triggered by a vaccine.
- End of a long road -
Sanofi and GSK developed the jab at the same time that they are waiting for regulatory approval for their first-generation vaccine.
The approval marks the end of a long journey for Sanofi to bring a Covid vaccine to market. The French pharma giant, considered to be a world leader on vaccines, has come under huge scrutiny at home for failing to roll out a Covid jab earlier.
The firm vowed to produce a billion vaccine doses in 2021, only for a dosage problem during clinical trials to send its researchers back to the drawing board. It also tried to develop a vaccine based on mRNA technology, only to abandon that plan as well.
While it struggled, Pfizer/BioNTech and Moderna brought their vaccines to market at a pace never before seen in history. Both vaccines were approved nearly two full years before Sanofi's breakthrough on Thursday.
"It is, it must be recognised, a failure... compared to the speed that was needed," Sanofi chairman Serge Weinberg told a shareholders' meeting in May.
While Sanofi has finally managed to get a Covid vaccine approved, the question remains about how much demand remains in an already crowded market.
Last week frontrunner Pfizer raised the annual sales forecast for its vaccine to $36 billion on the back of new deals for boosters.
Moderna meanwhile slashed the sales forecast for its own vaccine by $2-$3 billion dollars due to shipment delays.
On Thursday, French-Austrian biotech Valneva announced it will cut up to a quarter of its workforce.
Valneva became the first French firm to get a Covid vaccine approved by the EMA in June. It suspended production a month later, however, after the EU slashed its initial order of 60 million doses to just 1.25 million.
H.E.Young--AMWN


