-
 England Test captain Stokes to miss early county games in fitness battle
England Test captain Stokes to miss early county games in fitness battle
-
Macron vows to defend science as host of UN oceans summit

-
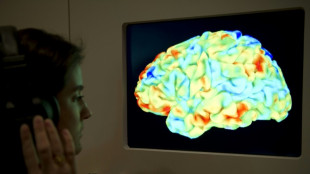 Brain implant turns thoughts into speech in near real-time
Brain implant turns thoughts into speech in near real-time
-
Top aide to Israel's Netanyahu arrested in 'Qatargate' probe

-
 Slashed US funding threatens millions of children: charity chief
Slashed US funding threatens millions of children: charity chief
-
China property giant Vanke reports annual loss of $6.8 bn

-
 World economies brace for Trump tariffs ahead of deadline
World economies brace for Trump tariffs ahead of deadline
-
Myanmar declares week of mourning as quake toll passes 2,000

-
 Japan leads hefty global stock market losses on tariff fears
Japan leads hefty global stock market losses on tariff fears
-
Yes, oui, Cannes! Glamour name eyes place in French Cup final

-
 'Different energy' at Man Utd after mini-revival, says Amorim
'Different energy' at Man Utd after mini-revival, says Amorim
-
Fear of aftershocks in Myanmar forces patients into hospital car park

-
 Far-right leaders rally around France's Le Pen after election ban
Far-right leaders rally around France's Le Pen after election ban
-
Renault and Nissan shift gears on alliance

-
 Hard-hitting drama 'Adolescence' to be shown in UK schools
Hard-hitting drama 'Adolescence' to be shown in UK schools
-
Primark boss resigns after inappropriate behaviour allegation

-
 Myanmar declares week of mourning as quake toll passes 2,000, hopes fade for survivors
Myanmar declares week of mourning as quake toll passes 2,000, hopes fade for survivors
-
Mbappe can be Real Madrid 'legend' like Ronaldo: Ancelotti

-
 Saka 'ready to go' for Arsenal after long injury lay-off: Arteta
Saka 'ready to go' for Arsenal after long injury lay-off: Arteta
-
Aston Martin to sell stake in Formula One team

-
 Three talking points ahead of clay-court season
Three talking points ahead of clay-court season
-
French court hands Le Pen five-year election ban

-
 Probe accuses ex J-pop star Nakai of sexual assault
Probe accuses ex J-pop star Nakai of sexual assault
-
Japan leads hefty global stock market losses on tariff woes

-
 Saka 'ready to go' after long injury lay-off: Arteta
Saka 'ready to go' after long injury lay-off: Arteta
-
Ingebrigtsen Sr, on trial for abusing Olympic champion, says he was 'overly protective'

-
 Tourists and locals enjoy 'ephemeral' Tokyo cherry blossoms
Tourists and locals enjoy 'ephemeral' Tokyo cherry blossoms
-
Khamenei warns of 'strong' response if Iran attacked

-
 France fines Apple 150 million euros over privacy feature
France fines Apple 150 million euros over privacy feature
-
UK PM urges nations to smash migrant smuggling gangs 'once and for all'

-
 Thai authorities probe collapse at quake-hit construction site
Thai authorities probe collapse at quake-hit construction site
-
France's Le Pen convicted in fake jobs trial

-
 Chinese tech giant Huawei says profits fell 28% last year
Chinese tech giant Huawei says profits fell 28% last year
-
Trump says confident of TikTok deal before deadline
-
 Myanmar declares week of mourning as hopes fade for quake survivors
Myanmar declares week of mourning as hopes fade for quake survivors
-
Japan's Nikkei leads hefty market losses, gold hits record

-
 Tears in Taiwan for relatives hit by Myanmar quake
Tears in Taiwan for relatives hit by Myanmar quake
-
Venezuela says US revoked transnational oil, gas company licenses

-
 'Devastated': Relatives await news from Bangkok building collapse
'Devastated': Relatives await news from Bangkok building collapse
-
Arsenal, Tottenham to play pre-season North London derby in Hong Kong

-
 Japan's Nikkei leads hefty equity market losses; gold hits record
Japan's Nikkei leads hefty equity market losses; gold hits record
-
Israel's Netanyahu picks new security chief, defying legal challenge

-
 Trump says US tariffs to hit 'all countries'
Trump says US tariffs to hit 'all countries'
-
Prayers and tears for Eid in quake-hit Mandalay

-
 After flops, movie industry targets fresh start at CinemaCon
After flops, movie industry targets fresh start at CinemaCon
-
Tsunoda targets podium finish in Japan after 'unreal' Red Bull move

-
 French chefs await new Michelin guide
French chefs await new Michelin guide
-
UK imposes travel permit on Europeans from Wednesday

-
 At his academy, Romanian legend Hagi shapes future champions
At his academy, Romanian legend Hagi shapes future champions
-
Referee's lunch break saved Miami winner Mensik from early exit


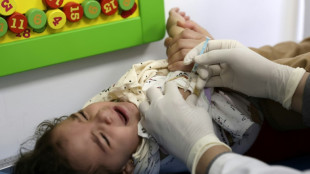
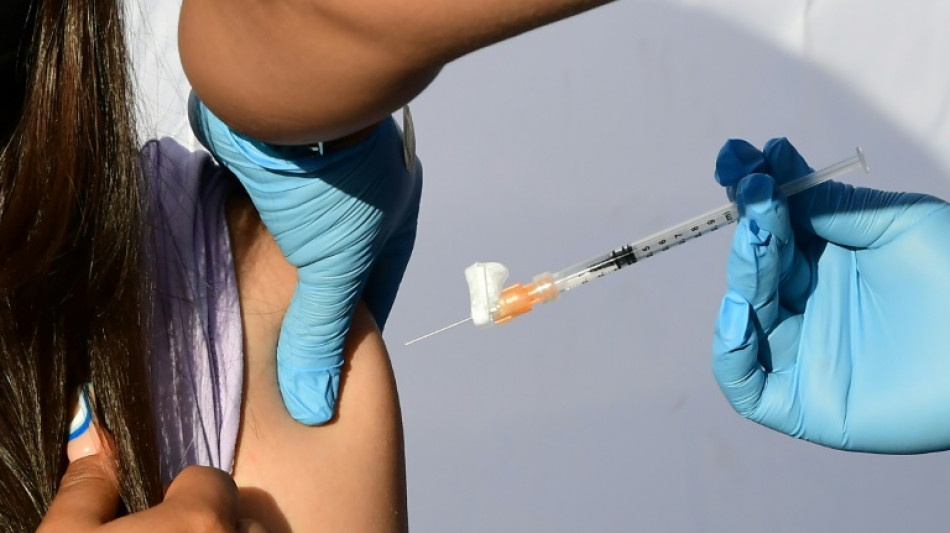
US panel weighs authorizing Covid vaccines for youngest children
After months of anxious wait for some parents, a panel of experts convened by the US Food and Drug Administration will meet Wednesday to weigh recommending Covid vaccines for the nation's youngest children.
Children under five are the only age group not yet eligible for Covid immunization in the United States and most other countries. If, as expected, panelists vote in favor of greenlighting the Pfizer and Moderna vaccines, formal authorizations should follow quickly, with the first shots in arms expected by next week.
Ahead of the meeting, the FDA posted its independent analyses of the two drugs, deeming both safe and effective. It also said there was a pressing unmet need to inoculate children in the age group since their rates of hospitalization and death "are higher than among children and adolescents 5-17 years of age."
Both vaccines are based on messenger RNA technology, which delivers genetic code for the coronavirus spike protein to human cells, training the immune system to be ready for when it encounters the real virus.
Pfizer is seeking authorization for three doses at three micrograms given to ages six months through four years, while Moderna has asked for the FDA to approve its vaccine as two doses of a higher 30 micrograms, for ages six months through five years.
They were tested in trials of thousands of children where they were found to have similar levels of mild effects to older age groups and triggered similar levels of antibodies.
Efficacy against infection was higher for Pfizer, with the company placing it at 80 percent compared to Moderna's estimates of 51 percent in the six-month-old to age two group and 37 percent in the two to five years age group.
But the figures are provisional and Moderna is studying adding a third dose later that may increase its figures.
There are some 20 million US children aged four years and under. If the FDA-appointed experts recommend the two vaccines, then the matter will go to another committee convened by the Centers for Disease Control and Prevention for a final say.
White House officials last week said the rollout of millions of shots at pharmacies and doctors' offices could begin as soon as June 21, following the Juneteenth holiday on June 20.
Of the total US Covid deaths, 480 have come in children under five, according to the latest official data.
Obesity, neurological disorders and asthma are associated with increased risk of severe disease, "however, a majority of children hospitalized for Covid-19 have no underlying medical conditions," the FDA said in a document.
Children can also go on to contract multisystem inflammatory syndrome in children (MIS-C), a rare but serious post-viral condition.
Data on long Covid in children is sparse, but the FDA's document cited a national survey in the United Kingdom which found that "among children ages two to 11 years who tested positive for COVID-19, 7.2 percent reported continued symptoms at 12 weeks."
L.Durand--AMWN