-
 Taiwan says US tariffs 'highly unreasonable'
Taiwan says US tariffs 'highly unreasonable'
-
Lawson says ruthless Red Bull axing was 'tough to hear'

-
 Heat humble Celtics for sixth straight win, Thunder roll on
Heat humble Celtics for sixth straight win, Thunder roll on
-
Trump escalates trade war with sweeping global tariffs

-
 Japan says US tariffs 'extremely regrettable', may break WTO rules
Japan says US tariffs 'extremely regrettable', may break WTO rules
-
South Koreans anxious, angry as court to rule on impeached president

-
 Juve at in-form Roma with Champions League in the balance
Juve at in-form Roma with Champions League in the balance
-
Injuries put undermanned Bayern's title bid to the test

-
 Ovechkin scores 892nd goal -- three away from Gretzky's NHL record
Ovechkin scores 892nd goal -- three away from Gretzky's NHL record
-
Australian former rugby star Petaia signs for NFL's Chargers

-
 China says opposes new US tariffs, vows 'countermeasures'
China says opposes new US tariffs, vows 'countermeasures'
-
Athletics world watching as 'Grand Slam Track' prepares for launch

-
 Heat humble Celtics for sixth straight win, Cavs top Knicks
Heat humble Celtics for sixth straight win, Cavs top Knicks
-
Quake-hit Myanmar's junta chief to head to Bangkok summit

-
 New Spielberg, Nolan films teased at CinemaCon
New Spielberg, Nolan films teased at CinemaCon
-
Shaken NATO allies to meet Trump's top diplomat

-
 Israel's Netanyahu arrives in Hungary, defying ICC warrant
Israel's Netanyahu arrives in Hungary, defying ICC warrant
-
Shiny and deadly, unexploded munitions a threat to Gaza children

-
 Stocks tank, havens rally as Trump tariffs fan trade war
Stocks tank, havens rally as Trump tariffs fan trade war
-
Altomare hangs on to tie defending champ Korda at LPGA Match Play

-
 Paraguay gold rush leaves tea producers bitter
Paraguay gold rush leaves tea producers bitter
-
Health concerns swirl as Bolivian city drowns in rubbish

-
 Syria says deadly Israeli strikes a 'blatant violation'
Syria says deadly Israeli strikes a 'blatant violation'
-
Financial markets tumble after Trump tariff announcement

-
 Starbucks faces new hot spill lawsuits weeks after $50mn ruling
Starbucks faces new hot spill lawsuits weeks after $50mn ruling
-
Europe riled, but plans cool-headed response to Trump's tariffs

-
 'Shenmue' voted most influential video game ever in UK poll
'Shenmue' voted most influential video game ever in UK poll
-
New coal capacity hit 20-year low in 2024: report

-
 Revealed: Why monkeys are better at yodelling than humans
Revealed: Why monkeys are better at yodelling than humans
-
Key details on Trump's market-shaking tariffs

-
 'A little tough love': Top quotes from Trump tariff talk
'A little tough love': Top quotes from Trump tariff talk
-
US business groups voice dismay at Trump's new tariffs

-
 Grealish dedicates Man City goal to late brother
Grealish dedicates Man City goal to late brother
-
US tariffs take aim everywhere, including uninhabited islands

-
 Trump sparks trade war with sweeping global tariffs
Trump sparks trade war with sweeping global tariffs
-
Israeli strikes hit Damascus, central Syria; monitor says 4 dead

-
 Slot 'hates' offside rule that gave Liverpool win over Everton
Slot 'hates' offside rule that gave Liverpool win over Everton
-
US stocks end up, but volatility ahead after latest Trump tariffs

-
 Barca oust Atletico to set up Clasico Copa del Rey final
Barca oust Atletico to set up Clasico Copa del Rey final
-
Mourinho grabs Galatasaray coach's face after losing Istanbul derby

-
 Grealish strikes early as Man City move up to fourth in Premier League
Grealish strikes early as Man City move up to fourth in Premier League
-
Reims edge out fourth-tier Cannes to set up PSG French Cup final

-
 Liverpool beat Everton as title looms, Man City win without Haaland
Liverpool beat Everton as title looms, Man City win without Haaland
-
Jota wins bad-tempered derby as Liverpool move 12 points clear

-
 Inter and Milan level in derby Italian Cup semi
Inter and Milan level in derby Italian Cup semi
-
Stuttgart beat Leipzig to reach German Cup final

-
 Trump unveils sweeping global tariffs
Trump unveils sweeping global tariffs
-
Italian director Nanni Moretti in hospital after heart attack: media

-
 LIV Golf stars playing at Doral with Masters on their minds
LIV Golf stars playing at Doral with Masters on their minds
-
Trump unveils sweeping 'Liberation Day' tariffs


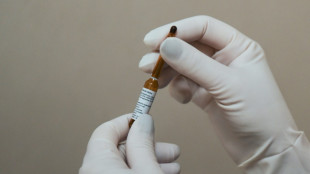
US issues world's first approval of RSV vaccine
The United States on Wednesday approved the world's first vaccine for the Respiratory Syncytial Virus (RSV), the culmination of a decades long hunt to protect vulnerable people from the common illness.
Drug maker GSK's Arexy was green-lighted for adults aged 60 and older, with similar shots from other makers including Pfizer and Moderna expected to follow soon.
"Today's approval of the first RSV vaccine is an important public health achievement to prevent a disease which can be life-threatening," said senior US Food and Drug Administration (FDA) official Peter Marks in a statement.
The decision "marks a turning point in our effort to reduce the significant burden of RSV," added Tony Wood, GSK's chief scientific officer.
RSV is a common virus that normally causes mild, cold-like symptoms, but can be serious for infants and the elderly, as well as those with weak immune systems and underlying conditions.
In severe cases it can cause pneumonia and bronchiolitis, an inflammation of the small airways deep inside the lungs.
According to the US Centers for Disease Control and Prevention, RSV leads to approximately 60,000 to 120,000 hospitalizations and 6,000 to 10,000 deaths among adults 65 years of age and older.
Awareness of the disease has increased in recent years, in part because of the strain it has placed on hospital systems over the last two winters.
Rates of RSV and flu fell during Covid-19 lockdowns, but surged when restrictions were lifted, with young children hit hard.
Pharmaceutical companies have been chasing an RSV vaccine for years. Given recent successful breakthroughs in the sector, analysts predict the market could be worth over $10 billion in the next decade, according to reports.
- More vaccines on way -
GSK's vaccine was approved based on a study of 25,000 people aged 60 and older that showed a single dose was 83 percent effective against disease caused by RSV, and more than 94 percent effective against severe disease.
Researchers will continue to follow volunteers in the study to assess the duration of protection as well as the safety and efficacy of more doses.
The most common side effects included injection site pain, fatigue, muscle pain, headaches and joint stiffness.
An irregular heartbeat was a less common side effect, occurring in 10 participants who received Arexy and four participants who received placebo.
Safety issues were also found in two other studies involving approximately 2,500 people aged 60 and up. In one of these studies, two volunteers developed a rare type of inflammation that affects the brain and spinal cord, and one of them died.
In the other study, one participant developed Guillain-Barré syndrome, in which the immune system damages nerve cells, causing muscle weakness and sometimes paralysis.
GSK's Arexy was recommended for approval last week by the European Union's drug watchdog, the European Medicines Agency, whose positive opinions are normally formally followed by approval from the European Commission.
Pfizer has said that it expects a decision from the FDA in May for its own RSV vaccine, also for those over 60 years old.
In January, Moderna said it hopes its RSV vaccine will be approved and available in time for the Northern Hemisphere's winter later this year.
Several other companies are also developing RSV vaccines.
Last year, the EU approved a preventative antibody treatment against RSV, developed by British-Swedish pharmaceutical firm AstraZeneca and France's Sanofi, which confers temporary protection.
D.Moore--AMWN