
-
 France detains alleged Romanian royal wanted in home country
France detains alleged Romanian royal wanted in home country
-
Van Dijk reveals 'progress' in talks over new Liverpool contract

-
 Starmer unveils support for tariff-hit auto sector
Starmer unveils support for tariff-hit auto sector
-
Clem Burke, drummer for Blondie, dies at 70

-
 Dortmund defender Schlotterbeck ruled out for season with injury
Dortmund defender Schlotterbeck ruled out for season with injury
-
Arteta says Arsenal can upset Real Madrid on 'biggest night' of career

-
 Bayern will not 'change goals' despite injury woes, says Kompany
Bayern will not 'change goals' despite injury woes, says Kompany
-
Inter captain Martinez fined 5,000 euros for blasphemy

-
 Netanyahu to plead with Trump for tariff break
Netanyahu to plead with Trump for tariff break
-
Arsenal's Saka says injury break 'really good' mentally

-
 EU funding of NGOs 'too opaque', auditors find amid political storm
EU funding of NGOs 'too opaque', auditors find amid political storm
-
La Liga appeal decision allowing Barcelona's Olmo to play again

-
 JPMorgan Chase CEO warns tariffs will slow growth
JPMorgan Chase CEO warns tariffs will slow growth
-
World sport-starved Moscow cheers Ovechkin NHL record

-
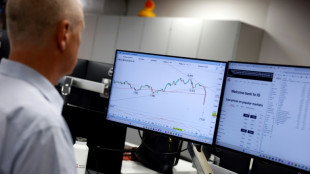 Stocks sink again as Trump holds firm on tariffs
Stocks sink again as Trump holds firm on tariffs
-
Trump warns against 'stupid' panic as markets plummet

-
 Thousands of Afghans depart Pakistan under repatriation pressure
Thousands of Afghans depart Pakistan under repatriation pressure
-
Macron rejects any Hamas role in post-war Gaza

-
 Boeing settles to avoid civil trial over Ethiopian Airlines crash
Boeing settles to avoid civil trial over Ethiopian Airlines crash
-
EU split on targeting US tech over Trump tariffs

-
 Russia, accused of stalling, wants answers before truce
Russia, accused of stalling, wants answers before truce
-
German climate activist faces expulsion from Austria after ban

-
 Southampton sack manager Juric after Premier League relegation
Southampton sack manager Juric after Premier League relegation
-
Fowler hits the target as Matildas down South Korea

-
 Brook named new England white-ball cricket captain
Brook named new England white-ball cricket captain
-
Honda executive resigns over 'inappropriate conduct'

-
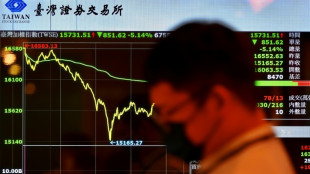 Stocks, oil prices sink further as Trump stands firm over tariffs
Stocks, oil prices sink further as Trump stands firm over tariffs
-
'Alarming' microplastic pollution in Europe's great rivers

-
 Spurs boss Postecoglou glad of Johnson revival ahead of Europa quarter-final
Spurs boss Postecoglou glad of Johnson revival ahead of Europa quarter-final
-
Major garment producer Bangladesh says US buyers halting orders

-
 Former Wales fly-half Biggar to retire at end of rugby season
Former Wales fly-half Biggar to retire at end of rugby season
-
African players in Europe: Iwobi ends goal drought to help sink Reds

-
 The worst market crashes since 1929
The worst market crashes since 1929
-
Japan emperor visits World War II battleground Iwo Jima

-
 'Everyone is losing money': Hong Kong investors rattled by market rout
'Everyone is losing money': Hong Kong investors rattled by market rout
-
China vows to stay 'safe and promising land' for foreign investment

-
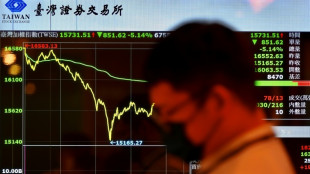 Stocks savaged as China retaliation to Trump tariffs fans trade war
Stocks savaged as China retaliation to Trump tariffs fans trade war
-
Unification Church appeals Japan's decision to revoke legal status

-
 Belgian prince seeks social security on top of allowance
Belgian prince seeks social security on top of allowance
-
European airlines hit turbulence over Western Sahara flights

-
 Boeing faces new civil trial over 2019 Ethiopian Airlines crash
Boeing faces new civil trial over 2019 Ethiopian Airlines crash
-
'Fear and anxiety': Bangkok residents seek quake-proof homes

-
 Injuries threaten to derail Bayern's home final dreams against Inter
Injuries threaten to derail Bayern's home final dreams against Inter
-
Real Madrid vulnerability evident ahead of Arsenal clash

-
 Texans warily eye impact of Trump's tariffs on their beloved trucks
Texans warily eye impact of Trump's tariffs on their beloved trucks
-
Equities savaged as China retaliation to Trump tariffs fans trade war
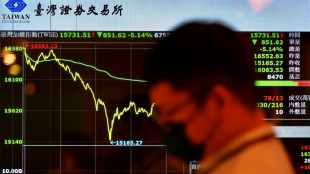
-
 Sara Duterte back in Philippines after month with detained father
Sara Duterte back in Philippines after month with detained father
-
Netanyahu and Trump to talk tariffs, Iran and Gaza

-
 Max power, Tsunoda's mixed debut, quick Kimi: Japan GP talking points
Max power, Tsunoda's mixed debut, quick Kimi: Japan GP talking points
-
Luis Enrique's revolution leaves PSG stronger without the superstars

| SCS | -3.23% | 10.249 | $ | |
| BCC | -3.72% | 92.015 | $ | |
| GSK | -6.33% | 34.355 | $ | |
| BTI | -1.5% | 39.27 | $ | |
| RIO | -1.59% | 53.815 | $ | |
| CMSD | -0.71% | 22.67 | $ | |
| CMSC | 0.04% | 22.3 | $ | |
| AZN | -5.68% | 64.78 | $ | |
| RBGPF | 1.48% | 69.02 | $ | |
| BCE | -4.17% | 21.8 | $ | |
| JRI | -4.55% | 11.439 | $ | |
| RYCEF | -0.61% | 8.2 | $ | |
| RELX | -7.05% | 44.99 | $ | |
| BP | -5.94% | 26.79 | $ | |
| NGG | -4.93% | 62.83 | $ | |
| VOD | -2.35% | 8.305 | $ |

CureVac Receives U.S. FDA IND Clearance to Initiate Phase 1 Clinical Trial for Novel mRNA-Based Precision Immunotherapy in Squamous Non-Small Cell Lung Cancer
Significant Regulatory Milestone: U.S. FDA cleared IND application for CVHNLC, CureVac's investigational therapy targeting squamous non-small cell lung cancer
Proprietary Epitopes: CVHNLC encodes novel tumor epitopes identified through proprietary whole genome-based discovery platform
Enhanced Combination Therapy: CVHNLC to be tested in combination with pembrolizumab, aiming to amplify targeted anti-tumor immune responses
Clinical Progress: Patient treatment anticipated to start in the second half of 2025
Pipeline Advancement: IND clearance highlights CureVac's continued oncology pipeline growth, with more candidates planned to enter the clinic in 2026
TÜBINGEN, GERMANY AND BOSTON, MA / ACCESS Newswire / April 7, 2025 / CureVac N.V. (Nasdaq:CVAC) ("CureVac"), a global biotech company developing a new class of transformative medicines based on messenger ribonucleic acid ("mRNA"), today announced that the U.S. Food and Drug Administration (FDA) has cleared its Investigational New Drug (IND) application for a Phase 1 clinical study of CVHNLC in patients with squamous non-small cell lung cancer (sqNSCLC). CVHNLC is CureVac's investigational mRNA-based precision immunotherapy consisting of two different mRNA constructs encoding eight tumor-associated antigens (TAAs) with prevalence across sqNSCLC patients. Encoded antigens include a novel class of TAAs that have not been previously tested in cancer immunotherapy trials.
The phase 1, dose-finding, open-label study will assess the safety and tolerability of CVHNLC plus pembrolizumab in patients with advanced sqNSCLC. The study comprises a dose-escalation part (Part A) of first-line maintenance treatment after either chemotherapy combined with pembrolizumab or pembrolizumab monotherapy. This is followed by an optional dose expansion part (Part B) in which CVHNLC is tested in combination with first-line chemotherapy and pembrolizumab.
"Immune checkpoint blockade has become a new standard of care for patients with metastatic squamous non-small cell lung cancer; however, overall prognosis still remains poor in advanced as well as in early settings of this disease, highlighting the urgent need for new therapeutic options," said Dr. Myriam Mendila, CureVac's Chief Scientific Officer. "Squamous non-small cell lung cancer exhibits a high prevalence of shared tumor antigens among patients, presenting a unique opportunity for developing targeted off-the-shelf mRNA immunotherapies. We believe by administering CVHNLC with checkpoint inhibition, we will trigger an amplified and targeted immune response, thereby increasing the efficacy against the cancer. Our goal is to take this combination also into earlier setting of the disease."
In Part A, patients with metastatic Stage IV sqNSCLC, who have received at least three cycles of pembrolizumab, either as monotherapy or in combination with chemotherapy, will be enrolled. CVHNLC doses between 100µg and 400µg plus pembrolizumab maintenance therapy for up to 12 months or until disease progression or undue toxicity occurs, will be administered with dose escalation. Primary endpoints include incidence of dose-limiting toxicities and treatment-related and emergent adverse events; secondary endpoints include overall response rate, progression-free survival, duration of response, and disease control rate.
"CVHNLC is our second oncology program to enter the clinical stage, highlighting the continued progress we are making with our mRNA-based precision immunotherapies. Importantly, we have been able to design CVHNLC using both known, shared tumor antigens and novel proprietary antigens discovered using our differentiated in-house technology platform," said Dr. Alexander Zehnder, Chief Executive Officer of CureVac. "We are leveraging this approach in the design of multiple novel cancer mRNA programs in our collaboration with MD Anderson Cancer Center, which we anticipate entering the clinic in the next 18-24 months."
About CVHNLC
CVHNLC has been built on CureVac's advanced second-generation mRNA backbone, featuring two different constructs utilizing unmodified mRNA formulated in lipid nanoparticles (LNPs). Its multi-epitope design encodes eight shared antigens, four of which are well-known with established relevance in solid tumors. The other four antigens are a novel class of TAAs uniquely derived from CureVac's proprietary whole-genome discovery platform.
About Squamous Non-Small Cell Lung Cancer
In the U.S., there are approximately 225,000 new cases of lung cancer each year, 87% of which are NSCLC, according to the American Cancer Society. Squamous non-small cell lung cancer (sqNSCLC) represents approximately 20-30% of all NSCLC cases and is considered a more aggressive form, posing significant challenges in disease control and treatment. Patients with sqNSCLC often face a tougher prognosis compared to other types of NSCLC. In the early sqNSCLC setting after neoadjuvant treatment, there is a 30-40% relapse rate within two years, with median overall survival of 15-17 months in metastatic setting.
About CureVac
CureVac (Nasdaq: CVAC) is a pioneering multinational biotech company founded in 2000 to advance the field of messenger RNA (mRNA) technology for application in human medicine. In more than two decades of developing, optimizing, and manufacturing this versatile biological molecule for medical purposes, CureVac has introduced and refined key underlying technologies that were essential to the production of mRNA vaccines against COVID-19, and is currently laying the groundwork for application of mRNA in new therapeutic areas of major unmet need. CureVac is leveraging mRNA technology, combined with advanced omics and computational tools, to design and develop off-the-shelf and personalized cancer immunotherapy product candidates. It also develops programs in prophylactic vaccines and in treatments that enable the human body to produce its own therapeutic proteins. Headquartered in Tübingen, Germany, CureVac also operates sites in the Netherlands, Belgium, Switzerland, and the U.S. Further information can be found at www.curevac.com.
CureVac Media and Investor Relations Contact
Dr. Sarah Fakih, Vice President Corporate Communications and Investor Relations
CureVac, Tübingen, Germany
T: +49 7071 9883-1298
M: +49 160 90 496949
[email protected]
Forward-Looking Statements of CureVac
This press release contains statements that constitute "forward looking statements" as that term is defined in the United States Private Securities Litigation Reform Act of 1995, including statements that express the opinions, expectations, beliefs, plans, objectives, assumptions or projections of CureVac N.V. and/or its wholly owned subsidiaries CureVac SE, CureVac Manufacturing GmbH, CureVac Inc., CureVac Swiss AG, CureVac Corporate Services GmbH, CureVac Belgium SA and CureVac Netherlands B.V. (the "company") regarding future events or future results, in contrast with statements that reflect historical facts. Examples include discussion of the potential efficacy of the company's vaccine and treatment candidates and the timing of programs entering the clinic. In some cases, you can identify such forward-looking statements by terminology such as "anticipate," "intend," "believe," "estimate," "plan," "seek," "project," "expect," "may," "will," "would," "could," "potential," "intend," or "should," the negative of these terms or similar expressions. Forward-looking statements are based on management's current beliefs and assumptions and on information currently available to the company. However, these forward-looking statements are not a guarantee of the company's performance, and you should not place undue reliance on such statements. Forward-looking statements are subject to many risks, uncertainties and other variable circumstances, including negative worldwide economic conditions and ongoing instability and volatility in the worldwide financial markets, ability to obtain funding, ability to conduct current and future preclinical studies and clinical trials, the timing, expense and uncertainty of regulatory approval, reliance on third parties and collaboration partners, ability to commercialize products, ability to manufacture any products, possible changes in current and proposed legislation, regulations and governmental policies, pressures from increasing competition and consolidation in the company's industry, the effects of the COVID-19 pandemic on the company's business and results of operations, ability to manage growth, reliance on key personnel, reliance on intellectual property protection, ability to provide for patient safety, fluctuations of operating results due to the effect of exchange rates, delays in litigation proceedings, different judicial outcomes or other factors. Such risks and uncertainties may cause the statements to be inaccurate and readers are cautioned not to place undue reliance on such statements. Many of these risks are outside of the company's control and could cause its actual results to differ materially from those it thought would occur. The forward-looking statements included in this press release are made only as of the date hereof. The company does not undertake, and specifically declines, any obligation to update any such statements or to publicly announce the results of any revisions to any such statements to reflect future events or developments, except as required by law.
For further information, please reference the company's reports and documents filed with the U.S. Securities and Exchange Commission (the "SEC"). You may get these documents by visiting EDGAR on the SEC website at www.sec.gov.
SOURCE: CureVac
View the original press release on ACCESS Newswire
D.Kaufman--AMWN