
-
 More work for McIlroy even with two wins before Masters
More work for McIlroy even with two wins before Masters
-
Trump hopeful of 'great' PGA-LIV golf merger

-
 No.1 Scheffler goes for third Masters crown in four years
No.1 Scheffler goes for third Masters crown in four years
-
Where Trump's tariffs could hurt Americans' wallets

-
 Trump says 'very close to a deal' on TikTok
Trump says 'very close to a deal' on TikTok
-
Trump tariffs on Mexico: the good, the bad, the unknown

-
 Postecoglou denies taunting Spurs fans in Chelsea defeat
Postecoglou denies taunting Spurs fans in Chelsea defeat
-
Oscar-winning Palestinian director speaks at UN on Israeli settlements

-
 With tariff war, Trump also reshapes how US treats allies
With tariff war, Trump also reshapes how US treats allies
-
Fernandez fires Chelsea into fourth as pressure mounts on Postecoglou

-
 South Korea court to decide impeached president's fate
South Korea court to decide impeached president's fate
-
Penguin memes take flight after Trump tariffs remote island

-
 E.T., no home: Original model of movie alien doesn't sell at auction
E.T., no home: Original model of movie alien doesn't sell at auction
-
Italy's Brignone has surgery on broken leg with Winter Olympics looming

-
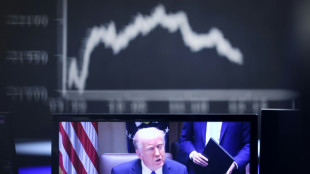 Trump defiant as tariffs send world markets into panic
Trump defiant as tariffs send world markets into panic
-
City officials vote to repair roof on home of MLB Rays

-
 Rockets forward Brooks gets one-game NBA ban for technicals
Rockets forward Brooks gets one-game NBA ban for technicals
-
Pentagon watchdog to probe defense chief over Signal chat row

-
 US tariffs could push up inflation, slow growth: Fed official
US tariffs could push up inflation, slow growth: Fed official
-
New Bruce Springsteen music set for June 27 release

-
 Tom Cruise pays tribute to Val Kilmer
Tom Cruise pays tribute to Val Kilmer
-
Mexico president welcomes being left off Trump's tariffs list

-
 Zuckerberg repeats Trump visits in bid to settle antitrust case
Zuckerberg repeats Trump visits in bid to settle antitrust case
-
US fencer disqualified for not facing transgender rival

-
 'Everyone worried' by Trump tariffs in France's champagne region
'Everyone worried' by Trump tariffs in France's champagne region
-
Italy's Brignone suffers broken leg with Winter Olympics looming

-
 Iyer blitz powers Kolkata to big IPL win over Hyderabad
Iyer blitz powers Kolkata to big IPL win over Hyderabad
-
Russian soprano Netrebko to return to London's Royal Opera House

-
 French creche worker gets 25 years for killing baby with drain cleaner
French creche worker gets 25 years for killing baby with drain cleaner
-
UK avoids worst US tariffs post-Brexit, but no celebrations

-
 Canada imposing 25% tariff on some US auto imports
Canada imposing 25% tariff on some US auto imports
-
Ruud wants 'fair share' of Grand Slam revenue for players

-
 Lesotho, Africa's 'kingdom in the sky' jolted by Trump
Lesotho, Africa's 'kingdom in the sky' jolted by Trump
-
Trump's trade math baffles economists

-
 Gaza heritage and destruction on display in Paris
Gaza heritage and destruction on display in Paris
-
'Unprecedented crisis' in Africa healthcare: report

-
 Pogacar gunning for blood and thunder in Tour of Flanders
Pogacar gunning for blood and thunder in Tour of Flanders
-
Macron calls for suspension of investment in US until tariffs clarified

-
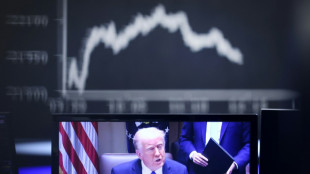 Wall St leads rout as world reels from Trump tariffs
Wall St leads rout as world reels from Trump tariffs
-
Mullins gets perfect National boost with remarkable four-timer

-
 Trump tariffs hammer global stocks, dollar and oil
Trump tariffs hammer global stocks, dollar and oil
-
Authors hold London protest against Meta for 'stealing' work to train AI

-
 Tate Modern gifted 'extraordinary' work by US artist Joan Mitchell
Tate Modern gifted 'extraordinary' work by US artist Joan Mitchell
-
Mexico president welcomes being left off Trump's new tariffs list

-
 Tonali eager to lead Newcastle back into Champions League
Tonali eager to lead Newcastle back into Champions League
-
Lesotho hardest hit as new US tariffs rattle Africa

-
 Stellantis pausing some Canada, Mexico production over Trump auto tariffs
Stellantis pausing some Canada, Mexico production over Trump auto tariffs
-
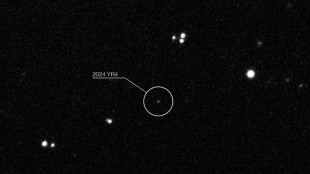
Rising odds asteroid that briefly threatened Earth will hit Moon
-
 Italy reels from Brignone broken leg with Winter Olympics looming
Italy reels from Brignone broken leg with Winter Olympics looming
-
Is the Switch 2 worth the price? Reviews are mixed


Hemogenyx Pharmaceuticals PLC Announces FDA Annual Report
Hemogenyx Pharmaceuticals Files Annual IND Report with FDA for HG-CT-1 CAR-T Therapy for AML
LONDON, UK / ACCESS Newswire / April 3, 2025 / Hemogenyx Pharmaceuticals plc (LSE:HEMO) is pleased to announce that it has submitted its Annual Report to the U.S. Food and Drug Administration (FDA) under the active Investigational New Drug (IND) application for HG-CT-1, the Company's proprietary CAR-T cell therapy for the treatment of relapsed or refractory acute myeloid leukemia (R/R AML).
The annual report provides a comprehensive update on the Company's activities under the IND during the first year of the clinical trial of HG-CT-1 and includes the following key elements:
1. Individual Study Information:
A summary of the ongoing clinical study, including its title, design, purpose and objectives, patient population, the total number of planned subjects, and the number enrolled as of the IND anniversary date (February 6, 2025). The report confirms the enrollment of the first patient and includes demographic details such as age, sex, and race.
2. Quality Summary Information:
Data obtained from the past year's investigations under the IND related to the stability of the HG-CT-1 drug product and the lentiviral vector used in its manufacturing.
3. Update to the General Investigational Plan:
A forward-looking update outlining the plan for continued patient enrollment during the upcoming year of the study.
This filing marks another important step in the Company's clinical development of HG-CT-1 and reaffirms its commitment to regulatory compliance and transparent communication with stakeholders.
Further updates will be provided as the trial progresses.
Dr. Vladislav Sandler, CEO & Co-Founder of Hemogenyx Pharmaceuticals, commented:
"Submitting our first annual IND report to the FDA is an important milestone that underscores the steady progress we are making in the clinical development of HG-CT-1. For patients facing relapsed or refractory AML, it brings us one step closer to delivering a potentially life-saving therapy. For our investors and partners, it demonstrates our continued execution, scientific rigor, and commitment to transparency. We are advancing with purpose and precision-guided by the urgency of patient need and the confidence of our supporters."
Enquiries:
Hemogenyx Pharmaceuticals plc | |
Dr Vladislav Sandler, Chief Executive Officer & Co-Founder | |
Peter Redmond, Director | |
SP Angel Corporate Finance LLP | Tel: +44 (0)20 3470 0470 |
Matthew Johnson, Vadim Alexandre, Adam Cowl | |
Peterhouse Capital Limited | Tel: +44 (0)20 7469 0930 |
Lucy Williams, Duncan Vasey, Charles Goodfellow | |
About Hemogenyx Pharmaceuticals plc
Hemogenyx Pharmaceuticals is a publicly traded company (LSE:HEMO) headquartered in London, with its US operating subsidiaries, Hemogenyx Pharmaceuticals LLC and Immugenyx LLC, located in New York City at its state-of-the-art research facility.
The Company is a clinical-stage biopharmaceutical group developing new medicines and treatments to treat blood and autoimmune disease and to bring the curative power of bone marrow transplantation to a greater number of patients suffering from otherwise incurable life-threatening diseases. Hemogenyx Pharmaceuticals is developing several distinct and complementary product candidates, as well as a platform technology that it uses as an engine for novel product development.
This information is provided by Reach, the non-regulatory press release distribution service of RNS, part of the London Stock Exchange. Terms and conditions relating to the use and distribution of this information may apply. For further information, please contact [email protected] or visit www.rns.com.
SOURCE: Hemogenyx Pharmaceuticals PLC
View the original press release on ACCESS Newswire
P.Santos--AMWN
